 
On the first test, the probability of YES is 1/1024, so the entropy of YES at that decision is. On the other hand, suppose you are doing linear search in a table of 1024 entries. By definition, the standard enthalpy of formation of an element in its most stable form is equal to zero under standard conditions, which is 1 atm for gases and 1 M for solutions. Here the entropy is used as a measure of how confident the classifier model is in its prediction. A thermodynamic systems enthalpy is defined as a. Use the data in Table 19.3.1 to calculate S° for the reaction of H 2 (g) with liquid benzene (C 6 H 6) to give cyclohexane (C 6 H 12 ). These values indicate that formation reactions range from highly exothermic (such as −2984 kJ/mol for the formation of P 4O 10) to strongly endothermic (such as +226.7 kJ/mol for the formation of acetylene, C 2H 2). The table below shows the standard enthalpy of formation, the standard Gibbs free energy of formation, standard entropy and molar heat capacity at constant pressure of several inorganic compounds. Entropy, on the other hand, is the amount of thermal energy in a system that cannot be converted into work. S° is positive, as expected for a combustion reaction in which one large hydrocarbon molecule is converted to many molecules of gaseous products. 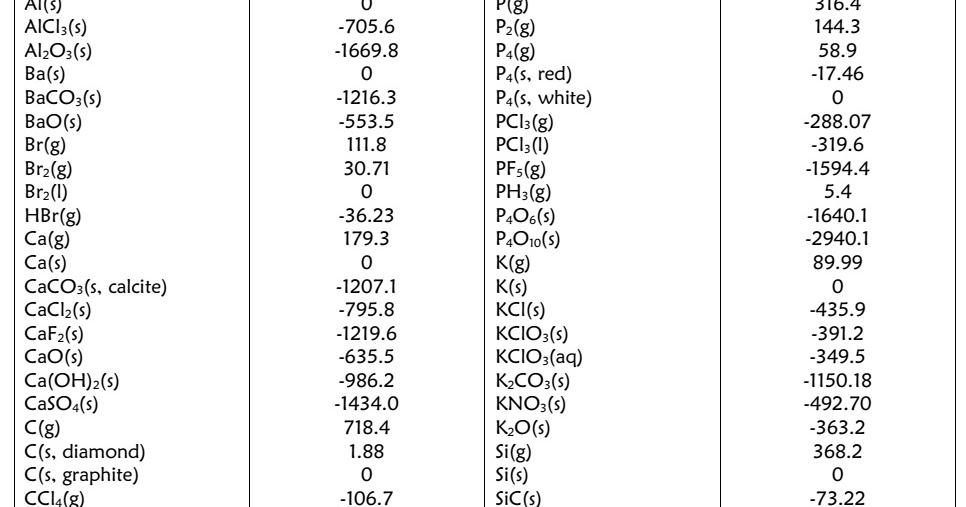
You will find a table of standard enthalpies of formation of many common substances in Tables T1 and T2. The following thermodynamic properties are calculated: density water, dynamic viscosity water, kinematic viscosity water, specific inner energy water, specific enthalpy water, specific entropy water, specific isobar heat capacity cp water, specific isochor heat capacity cv water, thermic conductivity water, speed of sound water. Inspired by industrial design, this set features a simple wooden-top table that gets a big boost in style. Entropy can be seen as a road map that connects thermodynamic situations, learn more about the first principles of entropy from this tutorial. An enthalpyentropy chart, also known as the HS chart or Mollier diagram, plots the total heat against entropy, describing the enthalpy of a thermodynamic. \) mole of N 2 and 1 mole of O 2 is correct in this case because the standard enthalpy of formation always refers to 1 mole of product, NO 2( g). astropy.table provides functionality for storing and manipulating heterogeneous tables of data in a way that is familiar to numpy users. Elevate the ordinary with the Entropy dining set. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
